Green Light from FDA, Children COVID-19 Vaccination May Start Soon?
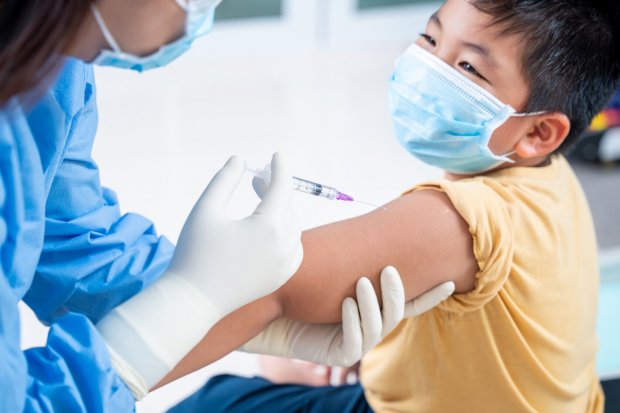
The US Food and Drug Administration (FDA) announced the clinical trial result of the Pfizer COVID-19 vaccine for children aged 5-11 years, showing that the vaccine has a 91 percent efficacy for the age group, VOA News reported. From the result, the US government's advisory medical panel agreed on October 26 to provide recommendations for authorizing Pfizer vaccines for children.
About 17 out of 18-panel members favored using Pfizer's vaccine for children, while another person decided to abstain. With the panel's decision, the FDA will grant permission to use Pfizer's vaccine for children. The government would immediately distribute the vaccine. "The government will ensure quick and equitable provision for all citizens," the US government said in a statement.

